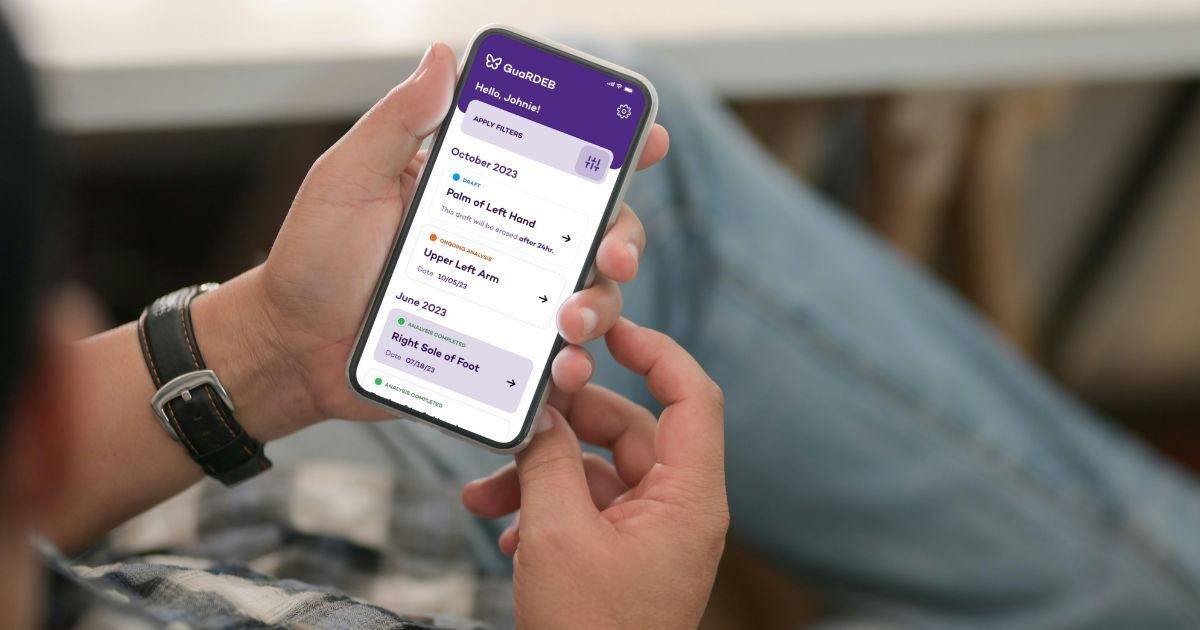
Enabling Rare-Disease Dermatology Research with a Privacy-First Web App
See how the Dermatology Department and the Institute for Artificial Intelligence in Medicine at Northwestern University partnered with LaunchPad Lab to build GuaRDEB, a secure, accessible web app that enables at-home image submission for consented RDEB participants and provides clinicians with a streamlined portal for research and longitudinal review.
The Client
The Dermatology Department and the Institute for Artificial Intelligence in Medicine at Northwestern University
Within the academic umbrella of Northwestern University Feinberg School of Medicine, Dr. Amy S. Paller (Chair, Department of Dermatology) and the Institute for Artificial Intelligence in Medicine (I.AIM), with funding from the EB Research Partnership, partnered on a project to modernize how consented, high-risk patients participate in image-based dermatology research, prioritizing accessibility, clinician review workflows, and responsible AI translation. I.AIM’s mission centers on bridging computational methods with human expertise to advance medical science and improve human health, with an explicit emphasis on responsible and ethical practice.
The Challenge
Transforming Urgent Dermatology Needs into Secure and Scalable Digital Workflows
Patients with Recessive Dystrophic Epidermolysis Bullosa (RDEB) have a high risk of aggressive cutaneous squamous cell carcinoma. In severe RDEB, it is a major cause of premature mortality. That reality makes timely monitoring and quick clinical review of lesions essential.
The Department of Dermatology and I.AIM teams were already developing strong AI methods to assess and categorize images, and the next step was to bring that research into a usable workflow for RDEB. They needed a real-world study platform that had to work for participants at home while also fitting into the research clinicians’ fast-paced review process.
Product Requirements
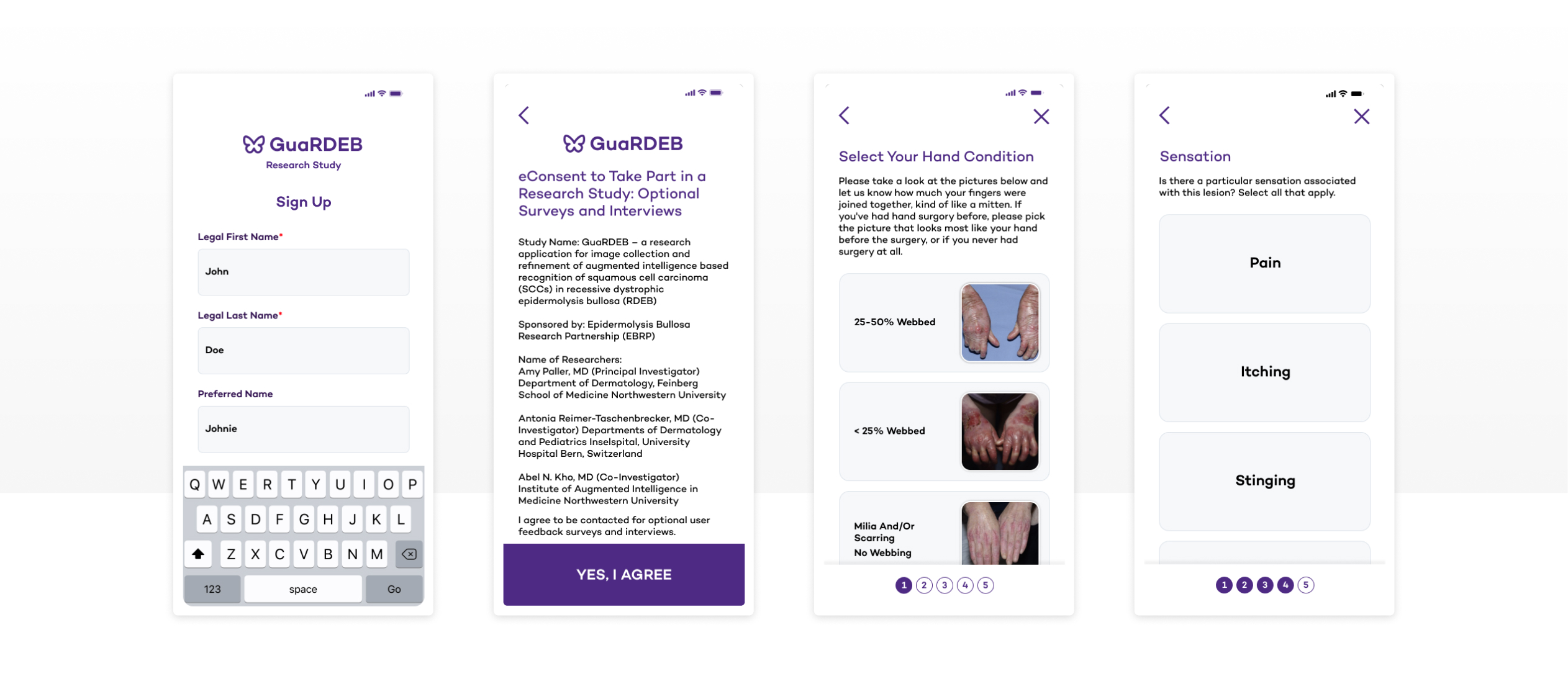
The Approach
Using Research to De-Risk Architecture, Privacy, and Accessibility
To move quickly without overcommitting, the team partnered with LaunchPad Lab on a structured Blueprint Workshop to set clear requirements, reduce risks early, and perform tight iteration loops.
Blueprint Workshop Outcomes
- Workflow Mapping: Enrollment, consent-informed study steps, image capture/upload, and clinician review queues
- Prototype Validation: Interaction patterns and end-to-end flow testing
- Technical Architecture: Web-first model integration patterns compatible with study operations
- Privacy & Security Planning: Stakeholder reviews appropriate for sensitive health data
- User Interface & Experience Testing: Accessibility-informed refinements to reduce participant friction
The output (prototype + architecture decisions) became the foundation for moving into a production build and strengthening funding readiness, all without positioning the prototype as a clinical diagnostic tool.
A Production-Ready Web App for Research Image Submission and Clinician Review
The solution was GuaRDEB—a Progressive Web App (PWA) built to streamline research participation and clinician review workflows for high-risk RDEB populations. Consented participants can securely submit wound images and brief context through any modern web browser, with a mobile-first experience optimized for quick phone capture and uploads. Clinicians and study staff use a dedicated portal to review, triage, and manage submissions within a consistent research workflow.
Modern Tech Stack
Leveraged React + Ruby on Rails, with Ionic to support a fast and reliable PWA experience.
Image Quality Pre-Checks
Automated validation to improve submission consistency and reduce rework and unnecessary processing.

Accessibility-Informed UX
Created design patterns shaped by usability testing to support participants with mobility limitations.
Clinician-Facing Review Portal
Implemented tools for organizing, tracking, and reviewing submissions for study operations.
The Results
Delivering Scalable Infrastructure for Research and Longitudinal Data Collection
The collaboration produced a platform optimized for what rare-disease research needs most: consistent, consented longitudinal data. Beyond enabling initial study enrollment and submissions, the system is designed to support repeat participation over time, creating a structured dataset that can help researchers study trends, support clinician review operations, and responsibly evaluate AI methods in real-world research settings.
Outcomes the Team Unlocked
Ready to Build Something Great?
Partner with us to develop technology to grow your business.
